For centuries, biology textbooks have drawn a hard line between humans and animals like salamanders: mammals heal with scars; amphibians regrow limbs. But new research from Texas A&M University suggests this distinction may be less about biological impossibility and more about a hidden switch that remains unflipped in human healing.
A study published in Nature Communications demonstrates that mice can regenerate bone, joints, ligaments, and tendons after amputation. The key was not introducing new stem cells, but rather redirecting the body’s existing cellular response away from scarring and toward regeneration.
While the regenerated tissues were not anatomically perfect, the findings challenge the long-held assumption that mammals have lost the ability to regrow complex structures. This breakthrough could pave the way for new treatments that reduce scarring and improve tissue repair, potentially bringing regenerative medicine closer to clinical reality.
Rewiring the Healing Response
When a mammal suffers a severe injury, such as an amputation, the body’s immediate priority is survival. Fibroblast cells rush to the wound site to seal it quickly, forming scar tissue. This process, known as fibrosis, protects the body from infection and blood loss, but it effectively shuts down the possibility of regrowing lost structures.
In contrast, regenerative animals like salamanders form a blastema —a temporary mass of cells that acts as a scaffold for new tissue growth.
Dr. Ken Muneoka, a professor at the Texas A&M College of Veterinary Medicine and Biomedical Sciences, spent his career investigating why this difference exists. His team discovered that mammalian fibroblasts are not inherently incapable of regeneration. Instead, they are trapped in a “scarring pathway” by default.
“It’s as if these cells can move in two different directions,” Muneoka explained. “They could either make a scar or make a blastema. Our research focused on redirecting the behavior of fibroblasts already present at the injury site.”
A Two-Step Treatment Strategy
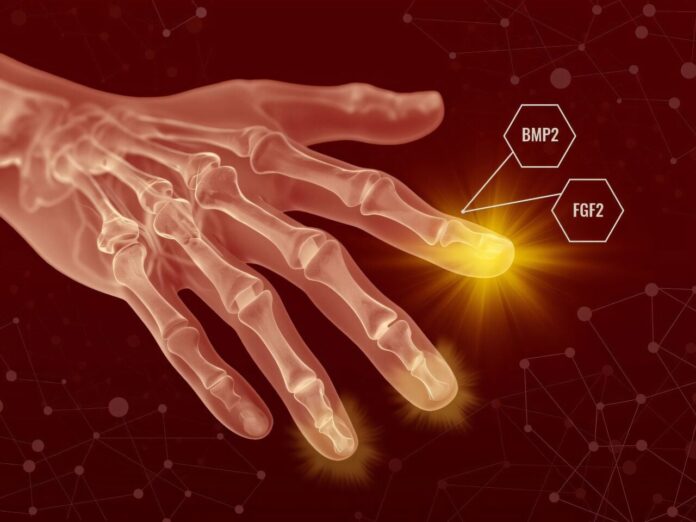
The researchers developed a sequential two-step treatment using two well-known growth factors to trick the body into regenerating tissue.
-
Step One: Prevent Scarring
After the initial wound closed, the team applied Fibroblast Growth Factor 2 (FGF2). This step allowed the body to finish its standard healing process but then signaled the fibroblasts to stop forming scar tissue and instead begin organizing into a blastema-like structure. -
Step Two: Build New Tissue
Several days later, the researchers applied Bone Morphogenetic Protein 2 (BMP2). This second signal instructed the newly formed cell mass to differentiate into specific tissues, including bone, tendon, and ligament.
“This is really a two-step process,” Muneoka said. “You first shift the cells away from scarring, and then you provide the signals that tell them what to build.”
Challenging the Stem Cell Paradigm
One of the most significant implications of this study is its challenge to current regenerative medicine strategies, which often rely on harvesting and transplanting stem cells.
Dr. Larry Suva, a co-author of the study, noted that the capacity for regeneration was not absent in mammals—it was merely obscured by the body’s default healing mechanism.
“The cells that we thought to be unprogrammable, in fact, are,” Suva said. “You don’t have to actually get stem cells and put them back in. They’re already there — you just need to learn how to get them to behave the way you want.”
The study also revealed a phenomenon called positional re-specification, where cells were guided to form structures outside their original anatomical position. This suggests that mammalian cells retain a flexible blueprint for development, which can be reactivated under the right conditions.
Imperfect but Functional Regrowth
The regenerated limbs in the mice were not perfect replicas of the original anatomy. The structures lacked the precise symmetry and fine detail of natural growth. However, they contained all the essential components removed during amputation: bone, joint structures, tendons, and ligaments.
“We regenerated what you would expect to see at that level of injury,” Muneoka said. “The structures are there — just not in a perfect form.”
This imperfection highlights the complexity of regeneration. It is not a simple on/off switch but a multifaceted process involving multiple biological pathways. While the results are not yet ready for human application, they provide a critical proof of concept: mammalian regenerative failure can be rescued.
Pathways to Clinical Application
While regrowing entire limbs in humans remains a distant goal, the immediate applications of this research may be more modest but equally impactful. The two-step approach could be adapted to reduce scarring and improve the quality of tissue repair in standard surgeries and injuries.
The clinical path forward may be smoother than anticipated because both growth factors are already familiar to the medical community:
* BMP2 is already FDA-approved for certain orthopedic procedures.
* FGF2 is currently undergoing multiple clinical trials.
Because these agents are not entirely new, regulatory hurdles may be lower, allowing researchers to test how slight shifts in the healing process can yield significant benefits in wound care and tissue repair.
Conclusion
This study marks a pivotal shift in our understanding of mammalian biology. By proving that the machinery for regeneration exists within us—dormant but accessible—researchers have opened new doors for medical innovation. The future of regenerative medicine may not lie in importing foreign cells, but in learning how to speak the body’s own language of repair.