On April 9, 1982, a scientific publication in the journal Science fundamentally altered our understanding of infectious diseases. Dr. Stanley Prusiner, then at the University of California, San Francisco, presented evidence that a disease could be caused not by a virus or bacteria, but by an infectious protein. This discovery challenged the very foundation of molecular biology and introduced the world to the prion.
The Mystery of the “Slow Virus”
For decades, scientists were baffled by a group of degenerative brain disorders that shared striking similarities. These included:
– Scrapie: A fatal disease affecting sheep and goats.
– Kuru: A devastating disorder observed in the Fore tribe of Papua New Guinea.
– Creutzfeldt-Jakob disease (CJD): A relentless human brain disorder.
Under a microscope, the brain tissue of victims of these diseases looked remarkably similar—riddled with tiny holes that gave the organ a “spongiform” or sponge-like appearance.
Because these diseases progressed slowly and lacked the typical hallmarks of infection—such as fever or immune system activation—scientists originally classified them as “slow viruses.” However, a massive logical gap remained: if these diseases were caused by viruses, they should contain nucleic acids (DNA or RNA). Furthermore, the fact that some cases appeared to run in families suggested a genetic component that didn’t align with standard viral transmission.
Challenging the “Central Dogma”
The breakthrough came when Prusiner pivoted his research from CJD to scrapie, prompted by findings from radiobiologist Tikvah Alper. Alper had demonstrated that scrapie could still be transmitted even after tissue was treated with ultraviolet light—a process that destroys DNA.
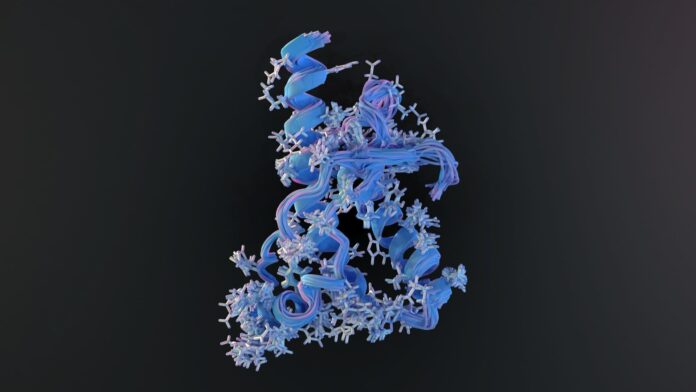
This observation suggested that the infectious agent lacked genetic material entirely. Through rigorous experimentation using hamsters (which accelerated the disease progression for study), Prusiner isolated the culprit. He proved that the infectious agent was a protein, which he named a “prion.”
This was a revolutionary claim because it contradicted the “central dogma” of molecular biology. The central dogma posits that information flows from DNA to RNA to protein; the idea that a protein alone could act as an infectious agent—essentially “coding” for its own replication by forcing healthy proteins to misfold—seemed impossible to the scientific establishment of the time.
How Prions Work: A Chain Reaction of Misfolding
The skepticism surrounding Prusiner’s work eventually gave way to overwhelming evidence. Over the following 15 years, researchers mapped out the mechanism of prion disease:
- Conformational Change: Prions are proteins that have folded into an abnormal, pathological shape.
- Template-Driven Infection: When a prion encounters a healthy version of the same protein, it acts as a template, forcing the healthy protein to “flip” into the diseased shape.
- Resistance: Unlike most proteins, prions are incredibly resilient and resist being broken down by the body’s natural degradation processes.
While prions drive the infection, modern science has also clarified the link to genetics: certain DNA mutations can predispose individuals to creating these misfolded proteins, explaining why some cases appear hereditary.
The discovery of the prion proved that life’s most fundamental rules could be bypassed by a single, misfolded molecule.
Conclusion
Stanley Prusiner’s identification of the prion bridged the gap between infectious disease and genetic pathology, earning him the Nobel Prize in 1997. His work transformed neurology and provided a new framework for understanding how proteins can act as agents of both life and death.